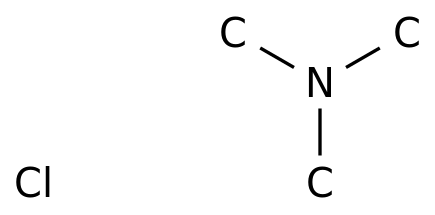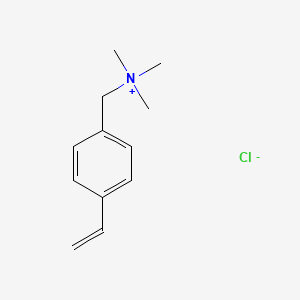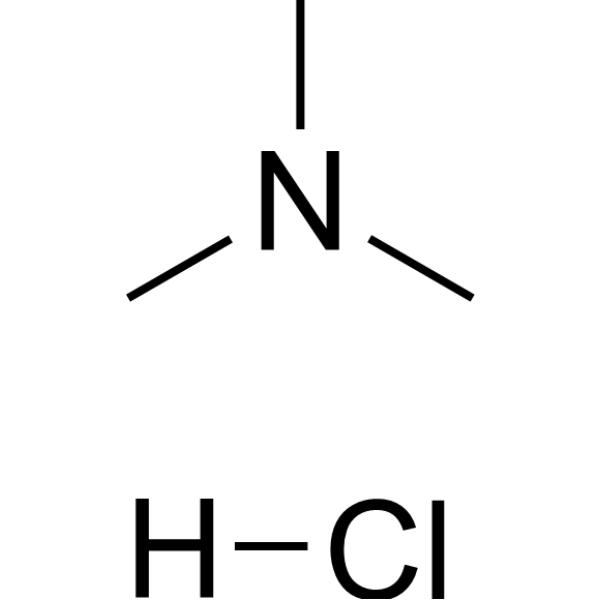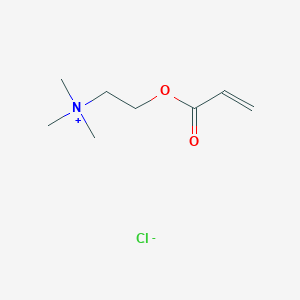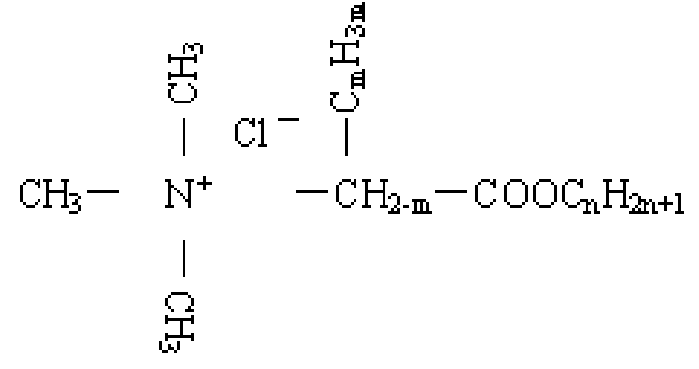
Carboxylic acid higher alcohol ester-trimethyl ammonium chloride and application thereof in clay stabilizer - Eureka | Patsnap develop intelligence library

Polymers | Free Full-Text | Study on Hydrolysis Properties and Mechanism of Poly(3-Methacrylamido Propyl Trimethyl Ammonium Chloride) Solution

![N-[3-(Trimethoxysilyl)propyl]-N,N,N-trimethylammonium chloride, 50% in methanol, N-[3-(Trimethoxysilyl)propyl]-N,N,N-trimethylammonium chloride, 50% in methanol,](https://www.thermofisher.com/TFS-Assets/CCG/Chemical-Structures/chemical-structure-cas-35141-36-7.jpg-250.jpg)





![Chemical structure of [2-(methacryloyloxy)ethyl] trimethylammonium... | Download Scientific Diagram Chemical structure of [2-(methacryloyloxy)ethyl] trimethylammonium... | Download Scientific Diagram](https://www.researchgate.net/publication/316749880/figure/fig1/AS:492283842830336@1494381022864/Chemical-structure-of-2-methacryloyloxyethyl-trimethylammonium-chloride-METAC.png)